Atomic Properties and Periodic Trends
|
Na Mg Ag Si P S Cl Ar |
Row 3
Ionization Energy (KJ/mol)
496 736 578 787 1012 1000 1251 1521 |
Atomic Radius (pm) 180 150 125 110 100 100 100 71 |
|
H Li Na K Rb Cs Fr |
Group 1
Ionization Energy (KJ/mol)
1312 520 496 419 403 376 393 |
Atomic Radius (pm) 25 145 180 220 235 260 270 |
1. Define atomic radius.
Atomic radius is half of the distance between radii in a covalently bonded diatomic molecule. It is used to measure the size of the cloud of the atom.
2. Use your graphs to describe the trends in atomic radius as you move across the row and down the group.
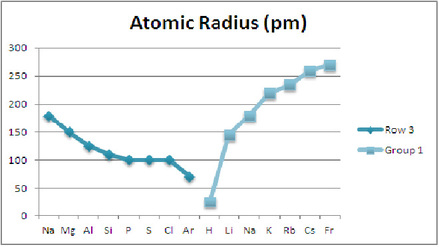
As the elements in periodic table move from left to right, the atomic radius decreases. The atomic radius decreases, because as the elements move across the row of periodic table, more protons (+) are present. The protons (+) attract the electron clouds (-) and shrink the overall size of the atom.
As the element in periodic table move from top to bottom, the atomic radius increases. The atomic radius increases, because as the elements move vertically in the periodic table, the energy level increases. The energy level makes the electron clouds bigger and increase the atomic radius.
The atomic radius is more impacted vertically than horizontally, because the energy levels of atoms are added as more and more electrons are added. The effect of protons that contracts the atom and its electron clouds more and more as more protons are added is not as significant as the increasing energy level; therefore, the atomic radius is more impacted vertically (increasing energy level) than horizontally (increasing number of protons).
In row 3, the Atomic Radius (pm) graph shows that the atomic radius, in pm, shrinks as the elements move from left to right.
In group 1, the Atomic Radius (pm) graph shows that the atomic radius, in pm, increases as the elements move from top to bottom.
As the element in periodic table move from top to bottom, the atomic radius increases. The atomic radius increases, because as the elements move vertically in the periodic table, the energy level increases. The energy level makes the electron clouds bigger and increase the atomic radius.
The atomic radius is more impacted vertically than horizontally, because the energy levels of atoms are added as more and more electrons are added. The effect of protons that contracts the atom and its electron clouds more and more as more protons are added is not as significant as the increasing energy level; therefore, the atomic radius is more impacted vertically (increasing energy level) than horizontally (increasing number of protons).
In row 3, the Atomic Radius (pm) graph shows that the atomic radius, in pm, shrinks as the elements move from left to right.
In group 1, the Atomic Radius (pm) graph shows that the atomic radius, in pm, increases as the elements move from top to bottom.
3. Explain the factors that determine these trends in atomic radius.
As explained in question number 2, the atomic radius is determined by energy level and the number of protons present.
Energy Level
Each of the atoms have electron clouds, a region of space where the electron fills up. There are energy levels that the electrons can fill, starting from level 1 to 7. Each energy levels can only have certain number of electrons occupying; in the first energy level, only 2 electrons can fit; in the second and subsequent energy levels, 8 electrons can fit. Once the electrons fill one energy level completely, it can start a new energy level and fill up with more electrons. As atom's electrons pile up in the electron clouds of the atom, the energy level of elements increase. Because electrons like to repel each other and occupy as many space as they can have, more energy level means more space that electrons take up, which increases the size of the electron cloud, and ultimately, increases the size of the entire atom itself. As elements move from top to bottom, energy level also increases, which explains why atomic radius also increases.
Protons
Energy levels usually decide which elements have bigger atomic radius than others, but when the compared elements have the same energy level, the size can be determined by observing the number of protons. First of all, the size, or the atomic radius, of the atoms is mainly made up of electron clouds, which consists of electrons. Electrons are negatively charged; in contrast, protons are positively charged. Because electrons and protons are oppositely charged, they naturally attract each other. Protons, which are located in the center of atoms, slightly pulls in electrons toward the center, slightly shrinking the size, or the atomic radius, of the atom. As more protons are present in the core, pulling attraction of electrons will increase and the cloud of electrons will slightly pull more and more into the center. This is why more atoms with more protons have smaller atomic radius than atoms with less protons, as long as the energy levels of the atoms are equal. Because the atoms on the right side of periodic table have more protons, they are smaller than the elements on the left side of periodic table; so atomic radii decrease as the elements move from left to right.
Energy Level
Each of the atoms have electron clouds, a region of space where the electron fills up. There are energy levels that the electrons can fill, starting from level 1 to 7. Each energy levels can only have certain number of electrons occupying; in the first energy level, only 2 electrons can fit; in the second and subsequent energy levels, 8 electrons can fit. Once the electrons fill one energy level completely, it can start a new energy level and fill up with more electrons. As atom's electrons pile up in the electron clouds of the atom, the energy level of elements increase. Because electrons like to repel each other and occupy as many space as they can have, more energy level means more space that electrons take up, which increases the size of the electron cloud, and ultimately, increases the size of the entire atom itself. As elements move from top to bottom, energy level also increases, which explains why atomic radius also increases.
Protons
Energy levels usually decide which elements have bigger atomic radius than others, but when the compared elements have the same energy level, the size can be determined by observing the number of protons. First of all, the size, or the atomic radius, of the atoms is mainly made up of electron clouds, which consists of electrons. Electrons are negatively charged; in contrast, protons are positively charged. Because electrons and protons are oppositely charged, they naturally attract each other. Protons, which are located in the center of atoms, slightly pulls in electrons toward the center, slightly shrinking the size, or the atomic radius, of the atom. As more protons are present in the core, pulling attraction of electrons will increase and the cloud of electrons will slightly pull more and more into the center. This is why more atoms with more protons have smaller atomic radius than atoms with less protons, as long as the energy levels of the atoms are equal. Because the atoms on the right side of periodic table have more protons, they are smaller than the elements on the left side of periodic table; so atomic radii decrease as the elements move from left to right.
4. How would the trend in ionic radii of period 3 and group 1 compare to the results you observed for atomic radii? Explain.
The trend in ionic radii of period 3 and group 1 compares to be similar.
Ionic radius is the radius of an atom's ion. Both ionic radii and atomic radii increase as the elements move from top to bottom and from right to left.
Ionic radius is the radius of an atom's ion. Both ionic radii and atomic radii increase as the elements move from top to bottom and from right to left.
5. Define first ionization energy.
Ionization energy is the minimum energy required to remove an electron from the outer shell of an atom. It is measured in KiloJoules per second and it differs with different elements.
6. Use your graphs to describe the trends in ionization energies as you move across the row and down the group.
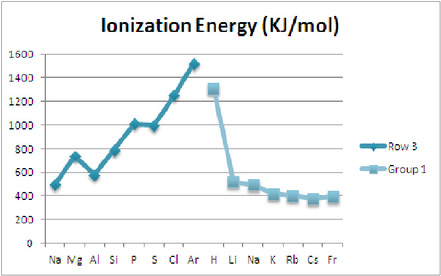
As seen in row 3, the graph shows that ionization energy usually increases as the elements move from left to right. The ionization energy increases as the elements move from left to right because the atomic radii of the elements decreases too. As the atoms become smaller, the electrons are closer to the protons in the core; it would take more energy to pull away an electron that is close to the protons than an electron farther away from the protons.
As seen in group 1, the graph shows that ionization energy usually decreases as the elements move from top to bottom. This also means that ionization energy usually increases as the elements move from bottom to top. This is true because of the atomic radii. If an atom is small, it takes more energy to pull away an electron from it because smaller atom has smaller distance between the electrons and protons, which makes the attraction between them stronger.
As seen in group 1, the graph shows that ionization energy usually decreases as the elements move from top to bottom. This also means that ionization energy usually increases as the elements move from bottom to top. This is true because of the atomic radii. If an atom is small, it takes more energy to pull away an electron from it because smaller atom has smaller distance between the electrons and protons, which makes the attraction between them stronger.
7. Explain the factors that determine these trends in ionization energies. Make sure you explain why the line is not smooth as you move across the row.
As explained in question 6, atomic radii determine the trends in ionization energies. It is easier for the electrons to leave its atom if the atom's size is bigger. Bigger atoms have higher energy levels, and higher energy levels lead to valence electrons being farther away from the protons in the core than the electrons in the inner energy levels. The electrons in the inner energy levels would block, or "shield", the attraction between proton and valence electrons, which explains why it is easier to take valence electrons away from a bigger atom than a smaller atom; this is called shielding effect. Therefore, ionization energy increases as the elements in the periodic table moves from left to right and from bottom to top because the size of the atoms shrinks as the elements move toward top right corner of the periodic table.
Although the size establishes the trends in ionization energy, some elements defy the trend. Ionization energy does not always increase as the elements move from left to right. This can be explained with a couple of explanations:
1. Orbitals- the electrons in p orbital is much easier to remove than electron in s orbital, because s orbital is closer to the protons than p orbital.
ex. Na- 496, Mg- 736, Al- 578, Si- 787
The ionization energy increases as the elements move from left to right, except at Al. Na and Mg are in s orbital, verses Al that is in p orbital. It is easier to remove electrons in p orbital than in s orbital, so Al is easier to remove and has lower ionization energy than Mg. Al is allowed to defy the trends of increasing ionization energy.
2. Electron Pairing- Repulsion between electrons are stronger in the same orbitals, so it would be easier to remove paired electrons than electrons with no pairs.
ex. Si- 787, P-1012, S- 1000, Cl- 1251, Ar- 1521
The ionization energy increases as the elements move from left to right, except S. At P, there would be 3 electrons in the p orbital with no pairs. (Hund's rule) The atom would like to spread the electrons out evenly, like so. When another electron is added to the p orbital, there would be 1 paired electron and 2 unpaired electron, which is uneven. The atom would rather have 3 unpaired electrons, so it would want to remove the electrons. This explains why S has lower ionization energy.
Although the size establishes the trends in ionization energy, some elements defy the trend. Ionization energy does not always increase as the elements move from left to right. This can be explained with a couple of explanations:
1. Orbitals- the electrons in p orbital is much easier to remove than electron in s orbital, because s orbital is closer to the protons than p orbital.
ex. Na- 496, Mg- 736, Al- 578, Si- 787
The ionization energy increases as the elements move from left to right, except at Al. Na and Mg are in s orbital, verses Al that is in p orbital. It is easier to remove electrons in p orbital than in s orbital, so Al is easier to remove and has lower ionization energy than Mg. Al is allowed to defy the trends of increasing ionization energy.
2. Electron Pairing- Repulsion between electrons are stronger in the same orbitals, so it would be easier to remove paired electrons than electrons with no pairs.
ex. Si- 787, P-1012, S- 1000, Cl- 1251, Ar- 1521
The ionization energy increases as the elements move from left to right, except S. At P, there would be 3 electrons in the p orbital with no pairs. (Hund's rule) The atom would like to spread the electrons out evenly, like so. When another electron is added to the p orbital, there would be 1 paired electron and 2 unpaired electron, which is uneven. The atom would rather have 3 unpaired electrons, so it would want to remove the electrons. This explains why S has lower ionization energy.
8. How would the trend in electron affinities of the elements in period 3 and group 1 compare to the results you observed for ionization energies? Explain.
The trend in ionization energy of period 3 and group 1 compares to be similar.
Electron affinity is an atom's ability to attract an atom. Both electron affinity and ionization energies increase when elements move from bottom to top and from left top right.
Electron affinity is an atom's ability to attract an atom. Both electron affinity and ionization energies increase when elements move from bottom to top and from left top right.
9. What are the characteristics of a metal, a nonmetal, a metalloid, and a noble gas?
Metals are good conductors of heat and of electricity. They are also solid, except for mercury, and have high melting points. They are malleable, or bendable. Most are shiny. They tend to lose electrons.
Nonmetals are not good conductors of heat and of electricity. They are generally in gaseous state, with some exceptions (bromine- liquid). They are not malleable. They are rarely shiny. They usually gain electrons.
Metalloids conduct heat and electricity better than nonmetals, but not as well as metals. They look metallic, but are not malleable. They take electrons from metals and lose electrons from nonmetals.
Noble gases are very unreactive. Their energy level is already full with electrons, so they will tend to neither gain nor lose electrons. They are odorless and colorless. They are in gaseous form in room temperature and are insoluble in water.
Nonmetals are not good conductors of heat and of electricity. They are generally in gaseous state, with some exceptions (bromine- liquid). They are not malleable. They are rarely shiny. They usually gain electrons.
Metalloids conduct heat and electricity better than nonmetals, but not as well as metals. They look metallic, but are not malleable. They take electrons from metals and lose electrons from nonmetals.
Noble gases are very unreactive. Their energy level is already full with electrons, so they will tend to neither gain nor lose electrons. They are odorless and colorless. They are in gaseous form in room temperature and are insoluble in water.
10. Classify the elements in period 3 as metals, metalloids, nonmetals, and noble gases.
Metals- Na, Mg, Al
Metalloids- Si
Nonmetals- P, S, Cl, Ar
Noble Gases- Ar
Metalloids- Si
Nonmetals- P, S, Cl, Ar
Noble Gases- Ar
11. Why are group 1 elements the most active metals and group 17 elements the most active nonmetals?
Both group 1 and group 17 are 1 electron away from having maximum number of electrons in the energy level. Atoms tend to gain or lose electrons to completely fill the spaces in the energy level. The atoms want the electrons to spread out evenly, according to Hund's Rule. Because group 1 and 17 are so close to having perfect amount of electrons, they are the most reactive out of all the groups.
Application of Ideas:
1. Create a periodic table to organize these elements based on properties. You may skip spaces.
2. Using your periodic table, predict the atomic number of an element with an atomic mass of 11.29 that has nonmetallic properties and is very reactive.
The atomic number of the reactive element with an atomic mass of 11.29 with nonmetallic properties is 5. Because the atomic mass number is 11.29, the element must lie between Delta (7.98) and Lambda (16.17). Because it has nonmetallic properties, it is on the right side of the periodic table. Because the element is very reactive, it cannot be on the far right or be a noble gas. So the most logical prediction of the atomic number is 5.
3. Predict the atomic number of an element having an atomic mass of 15.02 that exhibits metallic properties but is softer than lambda and harder than beta.
The atomic number of the element with an atomic mass of 15.02 that metallic properties and that is softer than lambda and harder than beta is 8. Because the atomic mass number is 15.02, the element must lie between Delta (7.98) and Lambda (16.17). Because it has metallic properties, it is on the left side of the periodic table. Because it is softer than lambda (group 3) and harder than beta (group 1), it must lie in group 2. Therefore, the atomic number of the element is 8.
4. Analyze your periodic table for periodic trends, and describe the trends that you identify.
On the left side, there are metals with metallic qualities. They are shiny, good conductors. They are also hard and dense. As elements move toward group 1 from the right, the elements become more reactive.
On the right side, there are nonmetals with nonmetallic qualities. They produce salt when combined with metal. They are also brittle and not malleable. As elements move toward group 17 from the left, the elements become more reactive.
As the elements go from right to left and top to bottom, the atomic radius increases.
As the elements go from left to right and bottom to top, the atomic radius decreases.
As the elements go from left to right and bottom to top, the ionization energy increases.
As the elements go from right to left and top to bottom, the atomic radius decreases.
On the right side, there are nonmetals with nonmetallic qualities. They produce salt when combined with metal. They are also brittle and not malleable. As elements move toward group 17 from the left, the elements become more reactive.
As the elements go from right to left and top to bottom, the atomic radius increases.
As the elements go from left to right and bottom to top, the atomic radius decreases.
As the elements go from left to right and bottom to top, the ionization energy increases.
As the elements go from right to left and top to bottom, the atomic radius decreases.
5. Which of these elements would have the largest atomic radius? The smallest?
Which of these elements would have the highest first ionization energy? The lowest?
Largest atomic radius- Beta
Smallest atomic radius- Delta
Highest first ionization energy- Delta
Lowest first ionization energy- Beta
Smallest atomic radius- Delta
Highest first ionization energy- Delta
Lowest first ionization energy- Beta

