Mole Ratio Lab
Purpose
To use experimental data to find the ratio of moles of reactants without using the formula of the reaction
Data Table
|
0.50 M of NaClO
0 mL 5.0 mL 10.0 mL 15.0 mL 20.0 mL 25.0 mL 30.0 mL 35.0 mL 40.0 mL 45.0 mL 47.0 mL 49.0 mL 50.0 mL |
0.50 M of Na2S2O3 in 0.20 M of NaOH
50.0 mL 45.0 mL 40.0 mL 35.0 mL 30.0 mL 25.0 mL 20.0 mL 15.0 mL 10.0 mL 5.0 mL 3.0 mL 1.0 mL 0 mL |
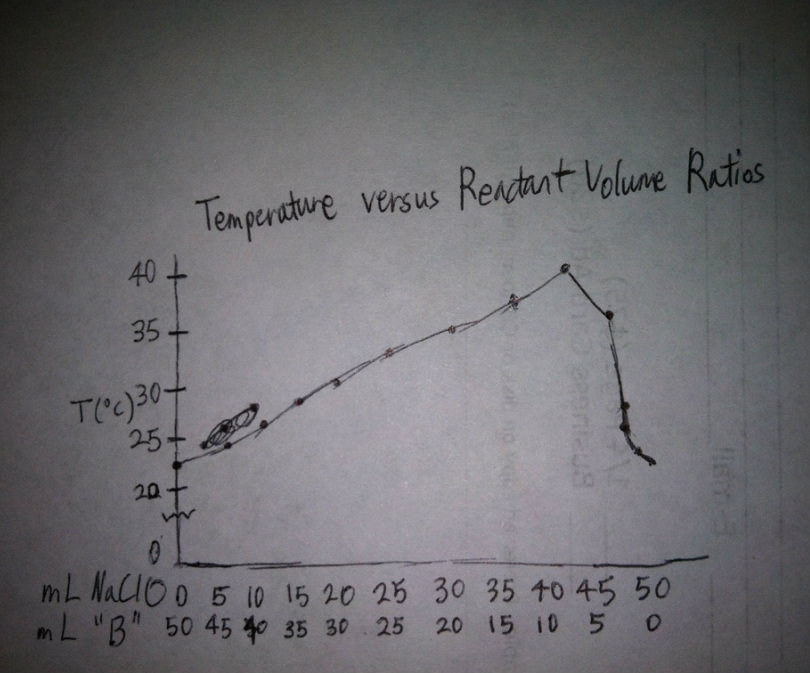
Temperature of the reaction (°C)
22.5 °C 24.5 °C 26.5 °C 28.5 °C 30.5 °C 32.5 °C 34.5 °C 36.5 °C 39.5 °C 35.0 °C 25.5 °C 23.0 °C 22.5 °C |
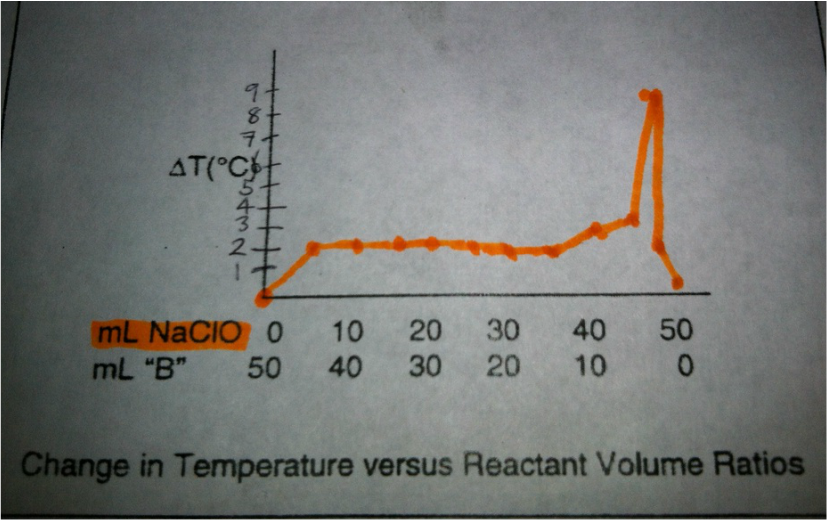
Changes in Temperature ΔT (°C)
0 °C 2 °C 2 °C 2 °C 2 °C 2 °C 2 °C 2 °C 3 °C 4 °C 9.5 °C 2.5 °C 0.5 °C |
Graph
Conclusion
The highest temperature of the reaction between two compounds corresponds with the accuracy of the optimum reactant volume ratios. The optimum mole ratio between NaClO and Na2S2O3 is 4 to 1.
Discussion of Theory
The Mole Ratio Lab uses the method of continuous variations to determine the mole ratio of two reactants without using the formula of the reaction. The method involves a series of experiments with different mole ratio of reactants each experiment to find the most accurate and productive ratio of two solutions. The optimum ratio, or the stoichiometric ratio, consume the greatest amount of reactants, form the greatest amount of product, and generate the most heat and maximum temperature change. The lab also highlights limiting reagents, a reactant compound with the least number of moles that limit the amount of product moles.
Analysis Questions
1. Why did you have to keep a constant volume of reactants?
In this lab, the data are measured to find the optimum ratio. To collect an accurate ratio between sodium hypochlorite and sodium thiosulfate, the solutions that are mixed together should have a different ratio for every trial. When the volume of the reactants is disturbed during the experiment, the ratio is also disturbed, making data unreliable and inaccurate.
2. What is meant by the term “limiting reagent”?
When making a cheeseburger, two buns, one patty, and one slice of cheese are required. If there are only seven buns, three patties, and two slices of cheese, the number of most cheeseburgers that can be made is two. The lack of cheese would limit the amounts of cheeseburgers that can be made, making the number of cheese the limiting factor. In the same way cheese limits the amount of cheeseburgers that can be made, limiting reagent can also limit the amount of products that react. Limiting reagent is a compound or element of the reactant that has the lowest number of moles to produce the product.
3. Which measurement limits the precision of your data: temperature or volume?
Precision only measures how close each data are. The measurement of temperature would limit the precision of the data, because even if the whole data was systematically inaccurate, the distance between each other would be correct and would be precise.
4. Which reactant is the limiting reagent along the upward sloping line of your graph? Which is the limiting reagent along the downward sloping line?
The limiting reagent along the upward sloping line of the graph is NaClO, because there are less grams of NaClO than grams of Na2S2O3. The limiting reagent along the downward sloping line of the graph is Na2S2O3, because there are less grams of Na2S2O3 than grams of NaClO .
5. What physical properties, other than temperature change, could use the method of continuous variations?
Using the method of continuous variations, mass of the precipitate that forms can be measured.
6. Why is it more accurate to use the point of intersection of the two lines to find the mole ratio, rather than the ratio associated with the greatest temperature change?
The point of intersection between the two line is necessary to find accurate the mole ratio, because it has at least three non-peak points in both sides of the peak.
In this lab, the data are measured to find the optimum ratio. To collect an accurate ratio between sodium hypochlorite and sodium thiosulfate, the solutions that are mixed together should have a different ratio for every trial. When the volume of the reactants is disturbed during the experiment, the ratio is also disturbed, making data unreliable and inaccurate.
2. What is meant by the term “limiting reagent”?
When making a cheeseburger, two buns, one patty, and one slice of cheese are required. If there are only seven buns, three patties, and two slices of cheese, the number of most cheeseburgers that can be made is two. The lack of cheese would limit the amounts of cheeseburgers that can be made, making the number of cheese the limiting factor. In the same way cheese limits the amount of cheeseburgers that can be made, limiting reagent can also limit the amount of products that react. Limiting reagent is a compound or element of the reactant that has the lowest number of moles to produce the product.
3. Which measurement limits the precision of your data: temperature or volume?
Precision only measures how close each data are. The measurement of temperature would limit the precision of the data, because even if the whole data was systematically inaccurate, the distance between each other would be correct and would be precise.
4. Which reactant is the limiting reagent along the upward sloping line of your graph? Which is the limiting reagent along the downward sloping line?
The limiting reagent along the upward sloping line of the graph is NaClO, because there are less grams of NaClO than grams of Na2S2O3. The limiting reagent along the downward sloping line of the graph is Na2S2O3, because there are less grams of Na2S2O3 than grams of NaClO .
5. What physical properties, other than temperature change, could use the method of continuous variations?
Using the method of continuous variations, mass of the precipitate that forms can be measured.
6. Why is it more accurate to use the point of intersection of the two lines to find the mole ratio, rather than the ratio associated with the greatest temperature change?
The point of intersection between the two line is necessary to find accurate the mole ratio, because it has at least three non-peak points in both sides of the peak.