Purpose
To find the unknown pH of the solution after a titration of HCl and NaOH.
Procedure
1. Wear safety goggles.
2. Measure 10mL of 1.5 HCl into a graduated cylinder and pour into a 50 mL oylemeyer flask.
3. Add two drops of phenolphthalein into the beaker of HCl.
4. Record your initial amout of NaOH.
5. Add NaOH until you start to see a color change. Swirl the flask as the color will be more difficult to swirl away. Faint pink is the desired color.
6. Dump out the neutralized solution, rinse out the flask and repeat twice.
7. Wash out the graduated cylinder, the oylemeyer flask, and the beaker. Clean-up the rest of the equipments.
2. Measure 10mL of 1.5 HCl into a graduated cylinder and pour into a 50 mL oylemeyer flask.
3. Add two drops of phenolphthalein into the beaker of HCl.
4. Record your initial amout of NaOH.
5. Add NaOH until you start to see a color change. Swirl the flask as the color will be more difficult to swirl away. Faint pink is the desired color.
6. Dump out the neutralized solution, rinse out the flask and repeat twice.
7. Wash out the graduated cylinder, the oylemeyer flask, and the beaker. Clean-up the rest of the equipments.
Data Table
Initial NaOH
45.3 mL
30.5 mL 18.7 mL 39.3 mL 28.7 mL |
Final NaOH
30.5 mL
18.7 mL 7.1 mL 28.7 mL 15.4 mL |
Changes in NaOH
14.8 mL
11.8 mL 11.6 mL 10.6 mL 13.3 mL |
pH
Basic
Basic Basic Basic Neutral |
Calculations
-Molarity of the NaOH
M1V1 = M2V2
n1 n2
HCl
1.5 M * 0.01 L = xM * 0.0133 L
1 mole 1 mole
x = 1.1 M NaOH
Th pH of the solutions after:
-No NaOH has been added.
[HCl] = [H+] = 1.5 M
pH = -log[1.5 M] = -0.18
-4.0 mL of NaOH has been added.
H+ + OH- <-> H2O
15 mmol 4.4 mmol
-4.4 mmol -4.4 mmol
10.6 mmol 0
pH = -log [10.6 mmol/14mL] = 0.12
-7.5 mL of NaOH has been added.
H+ + OH- <-> H2O
15 mmol 8.25 mmol
-8.25 mmol -8.25 mmol
6.75 mmol 0
pH = -log[6.75 mmol/17.5mL] = 0.41
-11.5 mL of NaOH has been added.
H+ + OH- <-> H2O
15 mmol 12.7 mmol
-12.7 mmol -12.7 mmol
2.3 mmol 0
pH = -log[2.3 mmol/21.5mL] = 0.96
-14.0 mL of NaOH has been added.
H+ + OH- <-> H2O
15 mmol 15.4 mmol
-15 mmol -15 mmol
0 0.4 mmol
pOH = -log[0.4 mmol/24mL] = 1.77
pH = 14 - 1.77 = 12.23
-18.0 mL of NaOH has been added.
H+ + OH- <-> H2O
15 mmol 19.8 mmol
-15 mmol -15 mmol
0 4.8 mmol
pOH = -log[4.8 mmol/28mL] = 0.17
pH = 14 - 0.17 = 13.23
M1V1 = M2V2
n1 n2
HCl
1.5 M * 0.01 L = xM * 0.0133 L
1 mole 1 mole
x = 1.1 M NaOH
Th pH of the solutions after:
-No NaOH has been added.
[HCl] = [H+] = 1.5 M
pH = -log[1.5 M] = -0.18
-4.0 mL of NaOH has been added.
H+ + OH- <-> H2O
15 mmol 4.4 mmol
-4.4 mmol -4.4 mmol
10.6 mmol 0
pH = -log [10.6 mmol/14mL] = 0.12
-7.5 mL of NaOH has been added.
H+ + OH- <-> H2O
15 mmol 8.25 mmol
-8.25 mmol -8.25 mmol
6.75 mmol 0
pH = -log[6.75 mmol/17.5mL] = 0.41
-11.5 mL of NaOH has been added.
H+ + OH- <-> H2O
15 mmol 12.7 mmol
-12.7 mmol -12.7 mmol
2.3 mmol 0
pH = -log[2.3 mmol/21.5mL] = 0.96
-14.0 mL of NaOH has been added.
H+ + OH- <-> H2O
15 mmol 15.4 mmol
-15 mmol -15 mmol
0 0.4 mmol
pOH = -log[0.4 mmol/24mL] = 1.77
pH = 14 - 1.77 = 12.23
-18.0 mL of NaOH has been added.
H+ + OH- <-> H2O
15 mmol 19.8 mmol
-15 mmol -15 mmol
0 4.8 mmol
pOH = -log[4.8 mmol/28mL] = 0.17
pH = 14 - 0.17 = 13.23
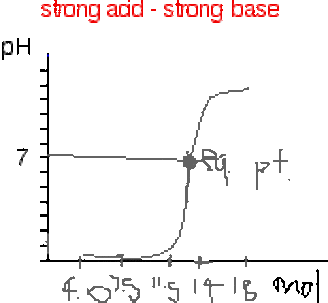
Graph
Conclusion
A titration was performed using 10.0 mL of 1.5 M HCl and appropriate amount of NaOH solution. Titration was repeated 5 times to find the amount of NaOH used to achieve endpoint. 14.8 mL, 11.8 mL, 11.6 mL, 10.6 mL, and 13.3 mL were used for each of the experiments. The average of the trial is 12.4 mL.
The molarity of NaOH was found by using the M1V1 = M2V2 equation, resulting in 1.1 M of NaOH.
The molarity of NaOH was found by using the M1V1 = M2V2 equation, resulting in 1.1 M of NaOH.
Discussion of Theory
In the Titrations Lab, 10.0 mL of 1.5 M HCl and appropriate amount of NaOH were titrated to find the molarity of NaOH and the pH of the solution after x mL of NaOH has been added. The lab discussed the difference between equivalence point, the point at which the reaction between titrant and unknown is complete, and the endpoint, the point where the indicator turns color. The color changes occurs when the concentration of more dominant form is ten times as great as the less dominant form in Ka = [In-][H+]/[HIn]. However, color changes in a solution does not necessarily equal to the equivalence point. Equivalence point can be found by observing the indicator, or using a pH meter and finding midpoint of vertical line in the titration curve. Endpoints can be found by observing the color change of the indicator.
Buffer solution was also discussed in this lab. Buffer solution is a solution that resists a change in pH when hydroxide ions or protons are added. It does so by reacting OH- with weak acid and H+ with conjugate base. Free OH- or H+ ions would not accumulate in the end.
The titration lab also involved indicators. Indicators are substances which undergoes a color change in the pH interval of the equivalence point, allowing physical observation of pH change. Most indicators are weak acids, so protons shift from acid to conjugate base. As the proton shifts, the color changes. The concentration of indicators in a solution do not change molarity value.
Buffer solution was also discussed in this lab. Buffer solution is a solution that resists a change in pH when hydroxide ions or protons are added. It does so by reacting OH- with weak acid and H+ with conjugate base. Free OH- or H+ ions would not accumulate in the end.
The titration lab also involved indicators. Indicators are substances which undergoes a color change in the pH interval of the equivalence point, allowing physical observation of pH change. Most indicators are weak acids, so protons shift from acid to conjugate base. As the proton shifts, the color changes. The concentration of indicators in a solution do not change molarity value.
Questions
1. What is the purpose of doing a titration?
To find the molarity of solutions, or NaOH in this lab.
2. What is an indicator (in detail!)?
Indicator shows the point where the reaction reaches the equivalence point, the point at which the reaction between titrant and unknown is complete. Color changes as the reaction reaches the equivalence point. Most are weak acids, so protons from weak acid would shift to conjugate base, causing the solution to change color.
3. How do you decide which indicator should be used for a titration?
Deciding which indicator to use depends on the types of titration. For strong acid-strong base titration, indicators with end points as far apart as pH 5 and pH 9 can be used. However, titration for weak acids or bases need carefully selected indicator with appropriate transition interval.
4. What is the difference between the equivalence point and the end point?
Equivalence point, also known as the stoichiometric point, is the point in a titration at which the reaction between titrant and unknown has been completed.
End point is the point in which the indicator turns color.
5. Compare and sketch a titration graph for a strong acid/strong base titration and for a weak acid/strong base titration.
See below.
6. Compare and sketch a titration graph for a strong acid/strong base titration and the same titration after a buffer solution has been added.
See below.
7. Explain what a buffer is and how a buffer solution keeps the pH from changing.
Buffered solution is a solution that resists a change in pH when either hydroxide ions or protons are added. It is solution of weak acids or bases containing a common ion. It contains either a weak acid and its salt, or a weak base and its salt.
When hydroxide ions or hydrogen ions are added to the solution, the ions do not accumulate.
OH- forms with weak acid to form conjugate base and water, leaving no OH- ions.
OH- + HA <-> A- + H2O
H+ forms with A- to form HA, leaving no H+ ions.
H+ + A- <-> HA
8. Consider each of the following potential sources of error. Answer:
"H" if it would have caused your calculated value for Molarity of NaOH to come out too high.
"L" if it would have caused it to come out too low.
"N" if it would have no effect on your value.
a) There was a little distilled water in the Erlenmeyer flask before you began the titration.- H
b) There was a little HCl in the Erlenmeyer flask before you began your titration.- L
c) You added 3 drops of phenolphtalein, insted of 2 drops.- N
d) An air bubble was present in the NaOH buret, but it stayed in while you titrated.- N
e) An air bubble was present in the NaOH buret, and it came out in the middle of your titration.- H
f) While you were titrating, some NaOH dripped onto te table, insteadof into the flask.- L
g) You forgot to add the phenolphthalein indicator.- H
To find the molarity of solutions, or NaOH in this lab.
2. What is an indicator (in detail!)?
Indicator shows the point where the reaction reaches the equivalence point, the point at which the reaction between titrant and unknown is complete. Color changes as the reaction reaches the equivalence point. Most are weak acids, so protons from weak acid would shift to conjugate base, causing the solution to change color.
3. How do you decide which indicator should be used for a titration?
Deciding which indicator to use depends on the types of titration. For strong acid-strong base titration, indicators with end points as far apart as pH 5 and pH 9 can be used. However, titration for weak acids or bases need carefully selected indicator with appropriate transition interval.
4. What is the difference between the equivalence point and the end point?
Equivalence point, also known as the stoichiometric point, is the point in a titration at which the reaction between titrant and unknown has been completed.
End point is the point in which the indicator turns color.
5. Compare and sketch a titration graph for a strong acid/strong base titration and for a weak acid/strong base titration.
See below.
6. Compare and sketch a titration graph for a strong acid/strong base titration and the same titration after a buffer solution has been added.
See below.
7. Explain what a buffer is and how a buffer solution keeps the pH from changing.
Buffered solution is a solution that resists a change in pH when either hydroxide ions or protons are added. It is solution of weak acids or bases containing a common ion. It contains either a weak acid and its salt, or a weak base and its salt.
When hydroxide ions or hydrogen ions are added to the solution, the ions do not accumulate.
OH- forms with weak acid to form conjugate base and water, leaving no OH- ions.
OH- + HA <-> A- + H2O
H+ forms with A- to form HA, leaving no H+ ions.
H+ + A- <-> HA
8. Consider each of the following potential sources of error. Answer:
"H" if it would have caused your calculated value for Molarity of NaOH to come out too high.
"L" if it would have caused it to come out too low.
"N" if it would have no effect on your value.
a) There was a little distilled water in the Erlenmeyer flask before you began the titration.- H
b) There was a little HCl in the Erlenmeyer flask before you began your titration.- L
c) You added 3 drops of phenolphtalein, insted of 2 drops.- N
d) An air bubble was present in the NaOH buret, but it stayed in while you titrated.- N
e) An air bubble was present in the NaOH buret, and it came out in the middle of your titration.- H
f) While you were titrating, some NaOH dripped onto te table, insteadof into the flask.- L
g) You forgot to add the phenolphthalein indicator.- H
Question 5

5. Compare and sketch a titration graph for a strong acid/strong base titration and for a weak acid/strong base titration.
Strong acid/strong base titration graph has the pH of 7 at the equivalence point.
Weak acid/strong base titration graph has higher pH than pH of 7 at the equivalence point.
Strong acid/strong base titration graph has the pH of 7 at the equivalence point.
Weak acid/strong base titration graph has higher pH than pH of 7 at the equivalence point.
Question 6

6. Compare and sketch a titration graph for a strong acid/strong base titration and the same titration after a buffer solution has been added.